Tetrahydropyridine
Forms of Tetrahydropyridines (abbreviated and often referred to generically by brewers as THP), specifically 2-acetyl-3,4,5,6-tetrahydropyridine and 2-acetyl-1,4,5,6-tetrahydropyridine (abbreviated ATHP or ACTPY), 2-ethyltetrahydropyridine (abbreviated ETHP or ETPY), and 2-acetylpyrroline (abbreviated ACPY or APY) [1], which are classified as a ketone and a cyclic imine [2], are commonly attributed to the "mousy", "urine" (in high amounts) Cheerios® or Cap'n Crunch® (in low amounts), "breakfast cereal", or more generically, "cracker biscuit" flavor in sour beers. The flavor is detected towards the end of the swallow, and the aftertaste can remain for a few minutes. Not all people are able to detect the flavor of THP. The low pH of sour beer or wine makes it harder to detect the flavor and often impossible to detect the aroma. An increase in pH is needed in order to detect the off-flavor, and the mouth's salivary pH serves that purpose when tasting beer or wine with THP. This effect on sensory detection by low pH might also explain why some people are better at detecting it since people have different pH's on the surface of their tongues and saliva [1]. Diacetyl is sometimes mistakenly indicated as a potential cause of this flavor in sour beers. However, Tetrahydropyridines are the accepted cause. The flavor tends to age out of sour beers after 2-6 months in the fermenter, kegs, or bottles (although aging periods as long as possibly 8-12 months have been reported on MTF [3]). The exact mechanism for how THP ages out of beer is not fully understood, and it is unknown whether cold or room temperature storage speeds up the breakdown process (it is more likely that room temperature storage will result in faster breakdown of THP) [4][5]. Many brewers have noticed that pitching rehydrated wine yeast at packaging reduces the amount/duration of this flavor in kegs and bottles [6].
In food, Tetrahydropyridines are associated with the aroma of baked goods such as white bread, popcorn, and tortillas, and is formed by Maillard reactions during heating.
Traditionally, the mousy/Cheerios® flavor from THP is considered an off flavor in both wine and sour beer. There is some debate and differing opinions as to whether or not a small amount of THP flavor is allowable (or even enjoyable) in sour beers, however most consider any level to be an off flavor.
Contents
History of Scientific Research
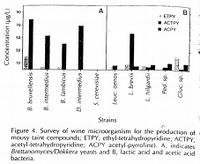
THP in wine ("mouse taint") was first described in wine by Müller-Thurgau and Osterwalder in 1913, although mention of an off-flavor in wine that 'is closely resembling to the smell of a residence of mice' dates to 1894 in "A Treatise on wines" by J.L.W. Thudichum. Müller-Thurgau and Osterwalder attributed it to being produced by bacteria. They also established that the ability to detect THP varies from person to person In the 1940's, some scientists proposed that THP was purely a chemical reaction, however their evidence was inconclusive. In 1977, Tucknott et al. was able to identify that THP production was attributed to both Brettanomyces yeast and lactic acid bacteria, and that ethanol and L-lysine played a role in its production, and this was confirmed by Heresztyn et al. in 1986 [7].
The chemical analysis of THP has proven to be a difficult endeavor in science. In 1995, Herderich et al. out of Australia developed a method for chemically analyzing THP for the first time. It was until this time that all three forms of THP (ETHP, ATHP, and APY) could be identified consistently in contaminated wine. It was also during this time that the three forms of THP contributed to the flavor of various foods, such as tortilla chips and taco shells. For example, APY was found in the fermentation of cocoa in 1995. In 1995, Grbin et al. described Brettanomyces yeast as the yeast that produces THP, while wine strains of S. cerevisiae did not [7].
In 2000, the Australians Grbin and Henschke showed that some strains but not all of B. bruxellensis, B. anomalus, B. nardenensis, and B custersianus produce THP, and that THP production was influenced by the carbon source, but not dependent on it (THP was still produced in dry wines with little available nutrients, and fermentation rate was not always related to THP levels) [8].
In 2007, the Australian team of Grbin et al. developed a complex and unique method of analyzing forms of THP using a Finnigan TSQ 70 mass spectrometer directly coupled to a Varian 3400 gas chromatograph. The chromatograph was equipped with a 30 m J&W Carbowax 20 CAM fused silica column, 0.25 mm i.d, and 0.25 μm film thickness. They were able to confirm that higher lysine levels increased the amount of ATHP produced (but not ETHP), although with diminishing increases of THP as the lysine level was increased. They also discovered that L-ornithine also functioned as a precursor for THP production in Brettanomyces. The group proposed a biochemical pathway for the different forms of THP in Brettanomyces [9].
Forms of THP
ATHP
ATHP/ACTPY (which has two forms: 2-acetyl-3,4,5,6-tetrahydropyridine and 2-acetyl-1,4,5,6-tetrahydropyridine) has a much lower flavor threshold than ETHP (see Thresholds), and has been historically cited as the cause of mousy off-flavors detected in wine. In wine, its aroma cannot be detected due to the low pH of wine (it can be detected if the pH is raised), only the flavor. It is easier to detect in higher pH wines. ATHP is the form of THP that is the major contributor to the aroma of freshly baked bread, corn tortilla chips, and crackers. How different foods/wines/beers interact with ATHP on the palate may explain the different flavors that are detected by people, as well differing concentrations and peoples' ability to detect ATHP [1].
ETHP
ETHP/ETPY (2-ethyl tetrahydropyridine) was first identified in wine in 1973, but until recently further studies weren't able to confirm its presence in wine. Its odor threshold is quite high (see Thresholds), and so it was not considered a major source of mousy off-flavors in wine for some time. Consequently, research on ETHP has been limited. More recently, it was shown that Lactic Acid Bacteria (LAB) can produce above threshold levels of ETHP, making it recently important to wine researchers [1].
It has been speculated by scientists studying mousy off-flavors in wine that its production is the result of slow metabolism of ATHP into ETHP by Brettanomyces. ETHP was observed to form much slower than ATHP, and coincided with a decrease in ATHP. This slow production of ETHP may be another reason it has been underestimated by researchers until recently [1]. ETHP has not been shown to break down into another byproduct.
APY
APY/ACPY (2-acetyl pyrroline) is a more volatile but more potent form of THP. It has a significantly stronger odor and lower odor threshold in wine than ATHP. It can also be found in damp pearl millet, white bread, taco shells, tortilla chips, and more aromatic rice such as Indian Basmati. APY is primarily produced by heterofermentative LAB. [1][7].
Unidentified "Transient" Forms
There have been anecdotal reports of other forms of mousy off-flavors. During growth of lactic acid bacteria (LAB), mousy off-flavor detection fluctuated with high levels detected early on, and lower levels detected towards the end of growth. This indicates that there may be a transient, strain-dependent form of THP that can occur during malolactic fermentation. There have also been sensory detection of mousy off-flavors at different levels than the documented levels of ATHP, ETHP, and APY, which were not associated with LAB or Brettanomyces [1].
Production

All species of Brettanomyces can produce forms of tetrahydropyridine in varying amounts, although some below threshold. Additionally, Lactic Acid Bacteria (LAB) including Lactobacillus and Pediococcus can produce forms of THP. Acetic Acid Bactera (AAB) has also been demonstrated to produce forms of THP [1][8].
Brettanomyces
Although the exact pathway is not known in Brettanomyces (several are proposed), the conditions for THP production are well documented. ATHP is produced by metabolizing the amino acid L-lysine or D-lysine [9], along with ethanol and a glucose or fructose molecule. Iron is also needed for THP production, although its exact role in biosynthesis is not known [1]. As with other amino acids, lysine is taken up by Saccharomyces during fermentation, and then released after fermentation. Levels of lysine fluctuate slightly throughout fermentation, but are generally high throughout a beer's lifetime (including after fermentation) [11][12][13][14]. Wheat generally has a slightly lower amount of lysine than barley, and oats have a slightly higher amount of lysine than barley [15][16][17][18].
Oxygen plays a key role and has a stimulatory effect in ATHP and ETHP production (particularly ATHP), but its exact role is not understood. It has been speculated that since ATHP production is associated with Brettanomyces growth, and Brettanomyces grows better under aerobic conditions, that this is why more ATHP is produced under aerobic conditions [19][9][20]. It has also been hypothesized that oxygen may have a direct effect on the THP molecules themselves [1]. ATHP production was also shown to increase when anaerobically precultured cells were transferred to an aerobic environment, indicating that oxygen has a direct role on the production of ATHP, not just a byproduct of Brettanomyces growth [1]. Limiting oxygen exposure during kegging/force carbonating is recommended for helping to reduce ATHP production; even very small amounts can have an effect (although the exact threshold of how much oxygen is required has not been determined). For example, the purity of the CO2 supply should thus be taken into consideration when force carbonating. At 0.5% impurity (the impurity is air, 1/5 of which is oxygen) and at 2 volumes of CO2, ~1,420 ppb of O2 would be added to the packaged beer, which is an exceedingly high amount of oxygen. The CO2 supply should ideally be 99.990% pure or better (this would introduce 46 ppb of oxygen at 2 volumes of CO2). The method that the CO2 is added can also determine how much oxygen is introduced into the packaged beer. Sparging CO2 (bubbling it through the beer) dissolves significantly less oxygen due to Henry's Law (see reference), while injecting (flushing) dissolves significantly more oxygen [21][22]. Vessel purging methods with CO2 are also less efficient than some might expect, and might still leave enough oxygen behind to stimulate THP production (see this HomebrewTalk thread). Pitching fresh Saccharomyces at bottling/kegging time and naturally carbonating the beer with sugar has reportedly reduced mousy off-flavor detection, perhaps because Saccharomyces metabolizes both the oxygen and sugar faster than Brettanomyces. Different strains of S. cerevisiae might be more efficient than others at helping reduce THP. For example, Mitch Ermatinger from Speciation Artisan Ales anecdotally observed that switching from CBC1 conditioning yeast to EC1118 reduced THP off-flavors from 1 month to two weeks [23] (see Packaging for details on re-yeasting at packaging time).
Interestingly, for unknown reasons Brettanomyces cells grown under aerobic conditions and then transferred to an anaerobic environment still produced significant amounts of ATHP in the anaerobic environment. It has been suggested that the aerobic conditions made the Brettanomyces cells predisposed to creating ATHP [1]. Oxygen exposure during Brettanomyces starters could potentially stimulate ATHP production later on down the road, even if the beer is not exposed to oxygen, although anecdotal evidence shows that this may not be a concern for brewers. It is still advised to use an aerobic or semi-aerobic starter for Brettanomyces unless the brewer believes this might be the direct cause of ATHP problems in their beer because Brettanomyces requires at least a small amount of oxygen for growth. Any other oxygen pick up after the beer has finished fermentation is the more likely cause of THP production and the brewer's post-fermentation processes should be examined first.
The level of ATHP production varies widely between species and strains of Brettanomyces, with some strains producing insignificant amounts and others producing very high amounts above taste threshold [8]. Additionally, ATHP production requires glucose or fructose, which explains why ATHP may be seen more often in stuck wine fermentations rather than wine that has finished fermenting. ATHP production by Brettanomyces was observed in wine with glucose or fructose added, along with synthetic growth media, suggesting that the type of growth substrate does not effect production [24].
The production of ATHP is not efficient, meaning that the amount of ATHP produced is not proportional to the amount of L-lysine consumed. Therefore, the production of ATHP appears to be a byproduct (secondary metabolite) of L-lysine catabolism [1]. ATHP is further metabolized into ETHP by Brettanomyces, although not much is known about this metabolic process [25][1]. ETHP has a significantly higher taste threshold, and is often not detected in contaminated wine [20].
Although Brettanomyces is capable of producing APY from L-ornithine [9], the amount produced is much less than that of LAB and high amounts of L-ornithine are required. In wine, there isn't enough L-ornithine present to production significant amounts of APY from L-ornithine. Therefore, the presence of APY (which is much easier to detect aromatically than ATHP) indicates a bacterial contamination in wine (it is unknown if this applies to beer) [1].
The presence of the "mousy off-flavor" caused by forms of THP appears to be temporary in beer. Although not much is known about the degradation or metabolic breakdown of ATHP/ETHP, it tends to age out of beer after 2-6 months. Since the odor/taste threshold for ETHP is much higher than ATHP, and ATHP appears to be metabolized into ETHP by Brettanomyces over time, this may be one of the mechanisms by which the mousy off-flavor ages out of beer. The possibility of ETHP breakdown is not mentioned in any studies that we know of. Another unknown is why does Brettanomyces produce ATHP shortly after kegging and force carbonating a beer that has reached final gravity. The most likely cause is oxygen pick up during the kegging process. Pitching fresh Saccharomyces at bottling/kegging time and naturally carbonating the beer with sugar has reportedly reduced mousy off-flavor detection, perhaps because Saccharomyces metabolizes both the oxygen and sugar faster than Brettanomyces.
Lactic Acid Bacteria
Heterofermentative Lactobacillus spp., particularly L. hilgardii and L. brevis, as well as Leuconostoc oeni [7], can also produce high levels of ATHP and APY from L-lysine/L-ornithine, ethanol (must be present), and iron. A strain of L. plantarum (L11a) was shown to produce relatively low amounts. L-lysine stimulates production of ATHP, and L-ornitine stimulates the production of APY [26][27][28][29][30][31]. Acetaldehyde has a stimulatory effect on ATHP and APY production, but is not required. No studies have been done to show whether or not oxygen plays a role in ATHP/APY production in LAB [1]. Most species of Pediococcus do not create forms of THP, although a few species do produce relatively small amounts. In particular, these include P. pentosaceus [32][33], and P. clausenii [34] (note that commercial cultures of Pediococcus are normally P. damnosus). Oenococcus oeni and Leuconostoc mesenteroides have also been associated with creating ATHP, APY, and ETHP all above threshold amounts. Since only heterofermentative species produce significant amounts of THP, it is thought that its production is linked to the heterolactic pathway, and thus the metabolism of sugars in LAB [26].
Acetic Acid Bacteria
Although research is limited, acetic acid bacteria (Gluconobacter sp. and many strains of Acetobacter aceti) have been shown to produce forms of THP [1].
Thresholds and Quantities Found in Mousy Wine
- Editor's note: the following thresholds are from studies on wine, and may not hold true for beer. As stated above, detection is influenced by pH, and so the low pH of sour beer may have the similar effect of repressing odor (more so) and taste (less so) detection, whereas non-sour Brettanomyces beers may have a higher detection rate.
- ETHP
- Odor threshold (wine): 150 µg/L
- Concentration reported in wines exhibiting mousy off-flavour: 2.7-18.7 µg/L (ETHP is generally not the cause of the detected mousy off-flavor)
- ATHP
- Odor threshold (water): 1.6 µg/L
- Concentration reported in wines exhibiting mousy off-flavour: 4.8-106 µg/L (ATHP is generally the cause of the detected mousy off-flavor)
- APY
Since a low pH hinders the detection of THP, one detection used by wine makers is to rub some wine on one's palm and smelling for THP. A more reliable method is to dip an alkaline paper strip into the wine, and then smelling the strip to detect the aroma of THP [7].
Discussions
What We Don't Know
- How does oxygen affect THP production biochemically?
- How much oxygen has an effect; is there a maximum amount of dissolved oxygen that would be insignificant in its production?
- Does bottle orientation during storage have an effect?
- Does creating an acid tolerance starter for conditioning yeast affect THP production?
- How do THP off-flavors age out of beer (hypothesis: ATHP is metabolized into ETHP by Brettanomyces, or somehow chemically degraded into ETHP)?
- Does storage temperature play a role in the degradation of THP off-flavors (cold vs room temp vs hot)?
- What genetic phenotypes determine high THP production in some strains of Brettanomyces and not others?
- What role does acetic acid bacteria play in THP production?
- What are the unidentified transient forms of THP, and do they apply to beer or just wine?
* Due to the specialized GC/MS equipment needed for measuring forms of THP that most labs do not have, certain answers will be difficult to obtain.
MTF Threads and Other Forum Posts
Below is a list of discussions on internet forum threads that may shed light on specific strains and individual experiences. Keep in mind that many of the opinions and experiences are anecdotal, although commonalities and shared experiences may prove to be useful and accurate.
- Joe Idnoni from House Cat Brewing and Brian Ogden from Attaboy Beer report that Caseinate de Potassium (a fining agent used in wine-making) was used to significantly reduce THP off-flavors in a kettle sour beer.
- MTF thread detailing experiences with certain strains and procedures (03/24/2017).
- Milk The Funk Thread on March 10, 2015.
- Milk The Funk Thread on Aug 25, 2015.
- Milk The Funk Thread on 09/17/2015.
- Homebrewtalk thread started by "ne0t0ky0".
- Homebrewtalk thread started by "loctones", comments by Michael Tonsmeire.
- Babblebelt thread with comments by Chad Yakobson.
- Michael Tonsmeire observes that adding fresh Saccharomyces at bottling time seems to reduce THP production, versus force carbonating.
- Kyle Weniger from Joseph James Brewing Co noticed significantly improved THP reduction storing a split batch of beer at room temperature versus storing cold.
- Chad Yakobson on The Sour Hour, 3/11/2015 (around 50 minute mark) - indicates that he has been researching THP for 2 years, and is continuing this research. Little else is mentioned.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 Mousy Off-Flavor: A Review. Eleanor M. Snowdon, Michael C. Bowyer, Paul R. Grbin, and Paul K. Bowyer. 2006.
- ↑ "6-Acetyl-2,3,4,5-tetrahydropyridine". Wikipedia. Retrieved 07/22/2016.
- ↑ MTF thread about how long THP takes to age out with comment by Mark Trent. 10/24/2016.
- ↑ MTF discussion regarding THP degredation under room temperature versus refrigeration temperatures. 10/28/2016.
- ↑ Tonsmeire, Michael. Homebrewtalk.com post 1. 11/21/2014. Retrieved 3/10/2015.
- ↑ Tonsmeire, Michael. Homebrewtalk.com post 2. 11/21/2014. Retrieved 3/10/2015.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 Developments in the sensory, chemical and microbiological basis of mousy taint in wine. Grbin, P.R, Costello, P.J, Herderich, M. 1996.
- ↑ 8.0 8.1 8.2 PAUL R. GRBIN and PAUL A. HENSCHKE. 2000.
- ↑ 9.0 9.1 9.2 9.3 The Role of Lysine Amino Nitrogen in the Biosynthesis of Mousy Off-Flavor Compounds by Dekkera anomala. Paul R. Grbin, Markus Herderich, Andrew Markides, Terry H. Lee, and Paul A. Henschke. J. Agric. Food Chem., 2007.
- ↑ Managing Wine Quality: Oenology and Wine Quality. A Reynolds Elsevier, Sep 30, 2010. Pg 359.
- ↑ The α-aminoadipate pathway for lysine biosynthesis in fungi. Hengyu Xu, Babak Andi, Jinghua Qian, Ann H. West , Paul F. Cook. Sept 2006.
- ↑ Lysine Biosynthesis in Saccharomyces cerevisiae: Mechanism of α-Aminoadipate Reductase (Lys2) Involves Posttranslational Phosphopantetheinylation by Lys5. David E. Ehmann , Amy M. Gehring , and Christopher T. Walsh. 1999.
- ↑ Elucidation of the Role of Nitrogenous Wort Components in Yeast Fermentation. C. Lekkas, G.G. Stewart, A.E. Hill, B. Taidi and J. Hodgson. May 2012.
- ↑ Proteins and amino acids in beers, their contents and relationships with other analytical data. S. Gorinstein, M. Zemsera, F. Vargas-Albores, J-L. Ochoa, O. Paredes-Lopez, Ch. Scheler, J. Salnikow, O. Martin-Belloso, S. Trakhtenberg. 1999.
- ↑ Amino Acid Composition of Six Grains and Winter Wheat Forage. Morey, D.D. 1983.
- ↑ "Oats". DIY Soylent website. Retrieved 02/07/2017.
- ↑ "Barley malt flour". DIY Soylent website. Retrieved 02/07/2017.
- ↑ "Wheat flour, whole-grain". DIY Soylent website. Retrieved 02/07/2017.
- ↑ Yakobson, Chad. The Brettanomyces Project; Introduction. Retrieved 3/10/2015.
- ↑ 20.0 20.1 Significance of Brettanomyces and Dekkera during Winemaking: A Synoptic Review. A. Oelofse, I.S. Pretorius, and M. du Toit. 2008.
- ↑ How the Purity of Sparged Carbon Dioxide Affects the Oxygen Concentration of Beer. Tap Into Hach blog. 01/24/2014. Retrieved 06/29/2017.
- ↑ How the Purity of Injected Carbon Dioxide Affects the Oxygen Concentration of Beer. Tap Into Hach blog. 12/02/2013. Retrieved 06/29/2017.
- ↑ Mitch Ermatinger. Milk The Funk Facebook group post on THP reduction using CBC1 and EC1118. 10/03/2017.
- ↑ Growth and volatile compound production by Brettanomyces/Dekkera bruxellensis in red wine. Romano A, Perello MC, de Revel G, Lonvaud-Funel A. J Appl Microbiol. 2008 Jun.
- ↑ Joseph, C.M. Lucy. Aromatic Diversity of Brettanomyces. U.C. Davis. Retrieved 3/10/2015.
- ↑ 26.0 26.1 Mousy Off-Flavor of Wine: Precursors and Biosynthesis of the Causative N-Heterocycles 2-Ethyltetrahydropyridine, 2-Acetyltetrahydropyridine, and 2-Acetyl-1-pyrroline by Lactobacillus hilgardii DSM 20176. Peter J. Costello and Paul A. Henschke. 2002.
- ↑ Formation of Substituted Tetrahydropyridines by Species of Brettanomyces and Lactobacillus Isolated from Mousy Wines. Tamila Heresztyn. 1986.
- ↑ Ability of lactic acid bacteria to produce N-heterocycles causing mousy off-flavour in wine. PETER J. COSTELLO1, TERRY H. LEE1, and PAULA. HENSCHKE. 2008.
- ↑ Sparrows, Jeff. Wild Brews. Brewers Publications. 2005. Pg. 112.
- ↑ Lahtinen, Ouwehand, Salminen, von Wright. Lactic Acid Bacteria: Microbiological and Functional Aspects, Fourth Edition. Pg 348.
- ↑ Heresztyn, Tamila. Formation of Substituted Tetrahydropyridines by Species of Brettanomyces and Lactobacillus Isolated from Mousy Wines.
- ↑ UniProt article. Retrieved 3/10/2015.
- ↑ UniProt article. Retrieved 3/10/2015.
- ↑ UniProt article. Retrieved 3/10/2015.
- ↑ Malolactic Fermentation 2005. Geneva on the Lake. Feb 2005. Retrieved 3/10/2015.