Difference between revisions of "Brettanomyces Storage Survival Experiment"
(Created page with "This page documents an experiment by [https://www.facebook.com/mark.trent.370?fref=nf Mark Trent] to investigate the survival rates or ''Brettanomyces'' under different media...") |
|||
| Line 1: | Line 1: | ||
| − | This page documents an experiment by [https://www.facebook.com/mark.trent.370?fref=nf Mark Trent] to investigate the survival rates or ''Brettanomyces'' under different media and temperatures. Further work is required to verify these results. Richard Preiss of [[Escarpment Laboratories]] is working on similar research, and Mark Trent intends on evaluating this experiment again. | + | This page documents an experiment by [https://www.facebook.com/mark.trent.370?fref=nf Mark Trent] to investigate the long term (100 days) survival rates or ''Brettanomyces'' under different media and temperatures. Further work is required to verify these results. Richard Preiss of [[Escarpment Laboratories]] is working on similar research, and Mark Trent intends on evaluating this experiment again. |
==Introduction== | ==Introduction== | ||
Revision as of 20:19, 26 June 2016
This page documents an experiment by Mark Trent to investigate the long term (100 days) survival rates or Brettanomyces under different media and temperatures. Further work is required to verify these results. Richard Preiss of Escarpment Laboratories is working on similar research, and Mark Trent intends on evaluating this experiment again.
Contents
Introduction
Survival of yeast in 4 different liquid media preparations was tested in cold and room temperature conditions. Media were; wort, buffered wort, MYPG, and buffered MYPG. In addition yeast was stored cold on solid media plates and slants containing buffered MYPG or MYPG. For liquid media, the yeast survived better at room temperature conditions compared to refrigerated conditions with the exception of survival in buffered MYPG cold treatment which was not significantly different from survival in buffered wort room or in buffered MYPG room (Fig. 2). Yeast stored on plates were dead on both media as was the yeast stored on MYPG slants while the yeast stored on buffered MYPG was viable in both replicates (Fig. 3).
Materials and Methods
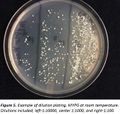
A strain of Brettanomyces isolated from Orval was inoculated into 4-250ml flasks containing either buffered MYPG, MYPG, buffered wort, or wort and incubated at 26°C on an orbital shaker at 80rpm for 60hrs. MYPG was prepared according to the methods of Chad Yakobson and was at a calculated gravity of 1.005. Wort was prepared from canned wort that had been mashed and cleared by traditional homebrew methods and was at a measured gravity of 1.04. For the buffered treatments, CaCO3 was added at 20g per L. Yeast cells from each culture were counted using a hemocytometer and 7ml aliquots of each culture were placed in a 15ml plastic centrifuge tubes for each treatment. Treatments were arranged in a completely randomized design and included Buffered MYPG-cold, Buffered MYPG-room, Buffered Wort-cold, Buffered Wort-room, MYPG-cold, MYPG-room, Wort-cold, and Wort-room. Cold treatments were held in a home style refrigerator at 1°C and room treatments were held in a closet that varied from 16 to 22°C. All treatments had 2 replicates. Data is represented as the percentage of live cell based on the original cell count of each inoculum source and the number of colonies that grew and were countable after dilution. The results were analyzed using Agstats02 (http://pnwsteep.wsu.edu/). After 100 days in storage, 100µl were sampled from each replicate and serial dilutions were made to 1:100000. Ten µl aliquots of the 1:100, 1:1000, and 1:10000 or 1:1000, 1:10000 and 1:1000000 dilutions were spread onto approximately 1/3 of a petri dishes containing MYPG solid media for each replicated treatment and subjected to 5 days incubation at 26°C (Fig. 5). Visible colonies were counted and the number of viable cells were calculated.
In addition, slants and plates were made by adding agar to the Buffered MYPG and MYPG media and were streak inoculated with the same strain of Brettanomyces. Plates were 100 x 15mm plastic petri dishes and slants were 15ml centrifuge tubes. Both plates and slants were incubated at 26°C until growth had covered the surface of the slants or 1mm colonies had formed on the plates. The plates were wrapped with electrical tape and the two replicates of each treatment were stored at cold temperature at 1°C. After 100 days in storage, a small amount of yeast from a single colony (plates) or spread (slants) was spread onto ½ of a petri dish containing MYPG solid media for each replicated treatment and subjected to 5 days incubation at 26°C. Only live/dead observations were made.
Results
Yeast grown for inoculum in liquid media appeared to grow at the same rate and reached similar numbers in the MYPG based media and the Wort based media. As expected the numbers were much lower in the MYPG based media with an averaged cell count of 222 million cells per ml. While the average of the wort based media was 483 million cells per ml (Fig. 1).
In general the yeast survived better at room temperature conditions compared to refrigerated conditions with the exception of survival in buffered MYPG cold treatment which was not significantly different from survival in buffered wort room or in buffered MYPG room. However survival in MYPG at room temperature and in wort at room temperature was significantly higher than in any other treatments with survival in the former 40% higher than in the latter. Yeast stored cold in buffered wort, MYPG and wort survived at or below 1 percent (Fig. 2).
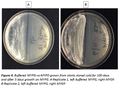
Yeast grown on buffered MYPG plates grew very slowly compared to those on MYGP plates. However, yeast grown on buffered MYPG slants grew at about the same rate as on MYPG slants (Fig.3). Viability of yeast stored cold on buffered MYPG and MYPG plates and slants was also evaluated after 100 days in storage. Yeast stored on plates were dead on both media as was the yeast stored on MYPG slants while the yeast stored on buffered MYPG was viable in both replicates (Fig. 4).
Limitations
The author only has access to a simple online statistical calculator that performs analysis of only randomized complete block or completely randomized experimental designs. This experiment should have been designed as a split plot. However I believe the statistics are relevant and reflect what was observed during the experiment. Good enough for MTF? Let the reader decide.
Survival in this experiment was determined by dilution plating. While this method has the advantage of proving the cell is viable by observing visual growth, it is known that yeast cells often clump together and are hard to separate. Therefore one cannot be certain the colony counted was derived from only one cell. Certainly the actual percentage of live cells was higher than indicated by the number of colonies observed.
Discussion
Previous experiments on survival of Brettanomyces by Richard Preiss and myself have shown the yeast survives better in cold temperatures up to 1 month. However, in my previous experiment both isolates seemed to have survived better at room temperature after 6 months although at a very low survival rate. The 6 months evaluation of that experiment had issues and therefore was repeated here. In Richards experiment, evaluation at 6 months revealed mixed results with one strain surviving better in cold temperature and the other better at warm temperature. Therefore, optimum survival temperature may vary by strain. In this experiment only one strain was used but clearly survived better after 100 days at room temperature in unbuffered MYPG. Buffering was deleterious to survival at room temperature in both media, while buffering MYPG for cold temperature was an advantage. It has long been known that Brettanomyces survives poorly on plates and in my experience poorly on slants. This experiment indicated that Brett survival on slants can be improved by buffering MYPG. Although the survival on slants was only tested as live or dead, the yeast on buffered MYPG grew up nicely after 100 days while the yeast on MYPG was dead. Since the yeast was dead on both buffered and unbuffered plates at 100 days, it is not known if buffering was an advantage for survival on plates.