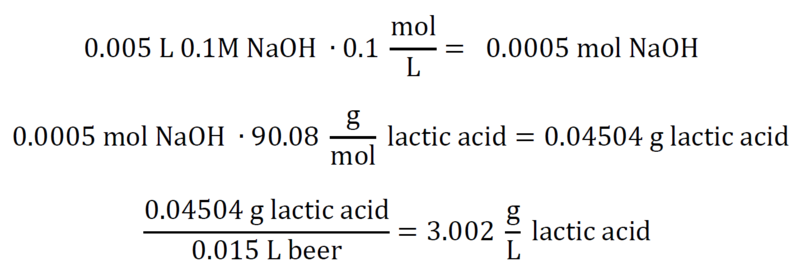Talk:Titratable Acidity
Example
"I agree that maths are hard." - Lance Shaner.
What you will need:
- A reliable and calibrated pH Meter.
- Sample of beer to be measured. Must be fully degassed if it has any carbonation (pour through a coffee filter, or shake and ventilate to decarbonate).
- Sodium Hydroxide, NaOH. Available in liquid or powder form. Be sure to note its molarity (M), units of mol/L. For more info on mol, see here.
Safety caution: always wear safety glasses and gloves when handling NaOH in any concentration. NaOH can cause severe burns. In concentrations higher than 0.1, NaOH can corrode through clothing. See Canadian Centre for Occupational Health and Safety on Sodium Hydroxide.
- Nitrile or latex gloves. NaOH is a strong base, it will hurt you if you get any on your skin.
- Pipettes and glassware, with precision down to 0.1 mL. Alternatively, you can use a precision scale to dose the base into the beer, if you know the density of both liquids (preferred method).
We need a precise volume of the beer. In this case, we have 15 mL. We also need NaOH in liquid form. Typically, it is sold in 0.1M form. Now, the trickiest part of this is adding precise amounts of NaOH (say, 0.1-0.5 mL at time), to your 15 mL of beer. Every time you add NaOH, you must vigorously stir the sample so it is well-mixed. Then you can measure its pH. You continue this until you reach the desired pH baseline of 8.2.
- Note: The baseline value of 8.2 pH is somewhat arbitrary, but it is the US (ASBC[1]) and Australian industry standard. A pH of 7 is a neutral pH and the pH of water, whereas ~8.2 is near the equivalence point for a lactic acid/sodium hydroxide reaction. A pH of 8.2 is also where a titration dye, phenolphthalein, changes color. A well-calibrated pH meter is easier to use than dye, not to mention its superior accuracy and precision, if used correctly (well-calibrated, probe well-maintained, etc.). A pH of 7 is the European industry standard for measuring TA in wine [2]. See also this MTF thread.
At or around a pH of 8.2, we have reached our equivalence point for a titration of pure NaOH and pure lactic acid. We need to convert the moles of NaOH we added into moles of lactic acid, and then divide the equivalent grams of lactic acid by the original volume of beer. That gets us g/L, and our titratable acidity. For a numerical example, assume 15mL beer, 5mL 0.1M NaOH:
The Eccentric Beekeeper TA Spreadsheet calculates TA as well as blends of beers with different TA values. It also includes a correction for beer final gravity. The idea is that the more residual sugar there is the less effect the acid will have on your perception. This is likely not that straightforward since you can have varying levels of sweetness at the same given FG [3].
In summary, the measurement of titratable acidity is a technique to quantify the total acid level of a beer. A major assumption was made: all the acid in the liquid was lactic acid. Two beers could have the same TA measurement, but have differing levels of palatable acidity, due to the acid makeup of the beer.
ASBC Modification Using Only Mass
The ASBC method requires both an accurate beer measurement volume and density. Depending on the quality of lab equipment available, these numbers can have varying error bars. In general, accurate scales are more affordable than accurate volume or density instruments; therefore, only using beer/acid mass will lead to a more consistent measurement.
For a numerical example, assume 15mL beer, 5mL 0.1M NaOH: