Nonconventional Yeasts and Bacteria
Nonconventional Yeasts and Bacteria are yeasts and bacteria genera that haven't been greatly explored in alcoholic fermentation, but might prove to be worth exploration. This page contains anecdotal information, as well as scientific information that might prove useful for brewers who are looking to brew with microbes that don't include the typical lab yeasts and bacteria for sour/mixed fermentations. For yeasts and bacteria that are more often used in sour and mixed fermentations, see Saccharomyces, Brettanomyces, Lactobacillus, and Pediococcus.
Under progress
Contents
Yeasts
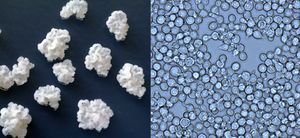
Zygosaccharomyces sp.
Zygosaccharomyces sp. belongs to the group of hemiascomycetous (class of fungi in which no ascocarps are formed) yeasts with a high tolerance to osmotic stress. This typical feature enables it to grow in environments with high concentrations of salts and/or sugars, i.e. under conditions restrictive to most other yeast species. Z. bailii, Z. bisporous, Z. rouxii, and Z. florentinus are species which have been isolated in grape musts or wine. Some strains can be very tolerant to a wide range of stressors, including 50% sugar, 2.5% acetic acid, 18% ethanol, and pH 2.0. It is also resistant to preservatives commonly used in beverage production such as SO2.
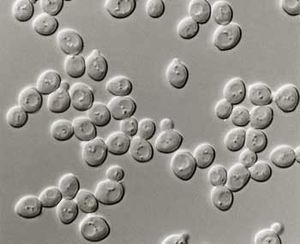
Toluraspora delbrueckii
Toluraspora delbrueckii is species of yeast, that is round to ovoid in shape and has been traditionally used in some wine fermentations to increase the complexity. Most of the commercial Torulaspora species and strains were isolated from soil, fermenting grapes (wine), berries, agave juice, tea-beer, apple juice, leaf of mangrove a tree, moss, lemonade and tree barks. Although it was said that most T. delbrueckii strains would not fully attenuate or tolerate higher alcohol contents it has been shown that this property is strain-dependent.
General Information
Analysis was done on 10 different T. delbruckii strains on various types of stress resistance as well as the ability to metabolize different carbon sources. The strains tested and the results are shown below. [1]
| Designation | Strain number/signature | Origin |
|---|---|---|
| T6 | RIBMa TdA | Wine |
| T9 | DSMb 70504 | Sorghum Brandy |
| T10 | CBSc 1146T | Unknown |
| T11 | TUMd 214 | Bottle (Pils beer, trace contamination, no beer spoilage observed) |
| T13 | TUMd TD1 | Wheat beer (starter culture) |
| T15 | TUMd 138 | Cheese brine |
| T17 | WYSC/Ge 1350 | Unknown |
| T18 | CBSc 4510 | Unknown |
| T19 | DSMb 70607 | Unknown |
| T20 | CBSc 817 | Unknown |
Hop Resistance
The resistance to alpha acids were also measured among these 10 strains using 0 PPM, 50 PPM, and 90 PPM. All strains were found to be resistant to these levels of alpha acids not affecting their growth. Some strains however were shown to have slower growth rates in the presence of 90 PPM and more. [1]
Ethanol Resistance
All 10 strains were also tested for their ability to grow in 5-10% ethanol content. The table below shows that all but one strain was able to grow in presence of 5% total alcohol but one thing they all shared in common is their inability to grow when in the presence of 10% alcohol. [1]
Growth (+) positive; (−) negative.
| Ethanol % | T6 | T9 | T10 | T11 | T13 | T15 | T17 | T18 | T19 | T20 |
|---|---|---|---|---|---|---|---|---|---|---|
| 5% | - | + | + | + | + | + | + | + | + | + |
| 10% | - | - | - | - | - | - | - | - | - | - |
Sugar Utilization
During fermentation trials of these 10 strains mentioned, sugar content was measured both before and after fermentation via HPLC. Tests showed the the sugar utilization of T. delbruekii is very strain dependent. All but one of the strains were shown to not ferment maltose and maltotriose. Although these tests do not show if these strains are able to utilize lactose, Eureka Brewing's blog mentions that they are unable to metabolize it.[2] The table below shows the percentages of sugars metabolized in the test wort by each strain. [1]
| Sugar Type | T6 | T9 | T10 | T11 | T13 | T15 | T17 | T18 | T19 | T20 |
|---|---|---|---|---|---|---|---|---|---|---|
| Fructose (%) | 93.2 | 92.3 | 91.5 | 88 | 91.6 | 90.2 | 84.5 | 96.4 | 93.6 | 88.1 |
| Glucose (%) | 96.6 | 96.2 | 97 | 96.6 | 97.3 | 95.5 | 94.5 | 95.4 | 94.7 | 89.6 |
| Sucrose (%) | 82.4 | 86.4 | 79 | 95 | 84.6 | 75.2 | 78.3 | 72 | 73.7 | 84.7 |
| Maltose (%) | 3.3 | 94.8 | 5.8 | 6 | 1.8 | 0.3 | 0.9 | 2.5 | 0.7 | 0.3 |
| Maltotriose (%) | 3 | 58.9 | 1.6 | 4.2 | .5 | 1.3 | 2.4 | 0.1 | 0.1 | 3.6 |
Cross Resistance
Again, all 10 strains growth was tested but this time with the presence of both 5% ethanol as well as 50 and 90 PPM of iso-alpha acid concentrations. Below you can see that with a combination of these two factors, growth was hindered in quite a few strains. [1]
Growth (+) positive; (−) negative.
| IBU/ethanol % (v/v) | T6 | T9 | T10 | T11 | T13 | T15 | T17 | T18 | T19 | T20 |
|---|---|---|---|---|---|---|---|---|---|---|
| 50/5 | - | + | - | + | - | + | + | + | + | - |
| 90/5 | - | - | - | + | - | + | + | + | + | - |
Wickerhamomyces sp.
See: Pichia sp.
Lachancea
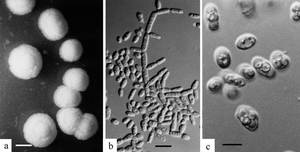
Pichia
Pichia is a genus of yeasts in the family Saccharomycetaceae with spherical, elliptical, or oblong cells. Pichia is a teleomorph, and forms hat-shaped, hemispherical, or round ascospores during reproduction. The anamorphs of some Pichia species are Candida species. The asexual reproduction is by multilateral budding. Pichia can be prolific pellicle-forming yeasts. [3]

Pichia kudriavzevii
P. kudriavzevii is a very abundant yeast found in soil, fruits, and various fermented beverages. It is ovoid to elongate in shape. So far, P. kudriavzevii is mainly associated with food spoilage to cause surface biofilms in low pH products. It is also known for known for creating a very heavy pellicle. [4]
Sugar Utilization
P. kudriavzevii can mainly metabolize glucose making it a non-viable strain for primary fermentations. During trials it was unable to metabolize galactose, sucrose, maltose, lactose, raffinose, and trehalose. [4] Interestingly, some strains of P. kudriavzevii can metabolize pentose sugars such as xylose [5].
Pichia apotheca
Pichia apotheca is a new hybrid species of Pichia which was identified in 2017. [6] Pichia apoteca was identified as a hybrid of Pichia membranifaciens and another unidentified species of Pichia.
Characterization
During the study, a fermentation using solely Pichia apoteca was conducted. The test wort used was at 13.75 degrees Plato and after 5 weeks at 13.61 degrees Plato. The alcohol by weight was found to be 0.02% after fermentation. In addition to this, percentages based on the entire contents of the wort showed that over five weeks, glucose levels dropped from 1.62% to 1.22%. The hybrid may incrementally breakdown maltotriose and fructose, dropping from 1.46% to 1.05% and 0.57% to 0.22% respectively, but did not appear to be able to reduce the levels of maltose. These results indicate that the Pichia hybrid did not significantly metabolize much of the available carbohydrates into alcohol within this wort environment. [6]
Pichia membranifaciens
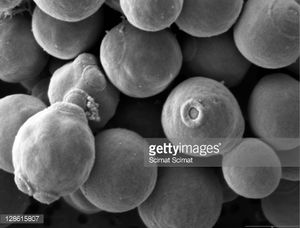
Debaryomyces sp.
Debaryomyces is a genus of yeast commonly referred to as a spoilage yeast [7]. The non-pathogenic species D. hansenii is commonly found in cheese, and is an osmotolerant, halotolerant, and xerotolerant (tolerant high amounts of salt and sugar, and low amounts of water) [8].
Debaryomyces hansenii
D hansenii is the most prevalent yeast in dairy and meat products as well as early stages of soy sauce fermentation. Various isolates exist originating from cheese, sake moto, edomiso, rennet, psoriasis, infected hands and salmon. In general, D. hansenii can be found in habitats with low water activity as well as in products with high sugar concentrations. Although D. hansenii is considered a non-pathogenic yeast, various clinical cases of D. hansenii exist. This yeast was originally isolated from saline environments and is maybe one of the most osmotolerant (can tolerate high levels of salt and sugar) yeasts in existence. [9]
General Information
As already mentioned, D. hansenii can tolerate very high levels of salt. Some sources cite salinity levels up to 24% whereas Saccharomyces cerevisiae commonly tolerate levels up to 10%. Such high tolerances are not that common in living organisms and can be used on industrial scale by cultivating D. hansenii at high salt levels to prevent the growth of other yeasts (quasi non-sterile production conditions). Beside dealing with high osmolarities, D. hansenii secrete toxins capable of killing other yeasts. [9]
Although this yeast is already an extremophile(an organism that thrives in physically or geochemically extreme conditions that are detrimental to most life on Earth) in terms of osmolarity, it does not stop there. Besides the normal sugars, D. hansenii is capable of metabolizing n-alkanes, melibiose, raffinose, soluble starch, inositol, xylose, lactic acid and citric acid. Furthermore, this yeast can form arabitol(a sugar alcohol) as well as riboflavin (vitamin B2). D. hansenii is therefore used on industrial scale to produce vitamin B2 and has a big potential for other biotechnological processes. [9]
D. hansenii is a very common yeast in cheeses and seems to have a major impact on the development of the microflora as well as the taste. As previously mentioned, D. hansenii can metabolize lactic acid, citric acid and galactose. The metabolization of lactic acid by yeasts has been shown to have an impact on the bacterial flora of the cheese in types such as Limburger, Tilsiter, Port Salut, Trappist, Brick and the Danish Danbo. Furthermore, D. hansenii forms volatile compounds associated with a “cheesy” flavor. For example, D. hansenii seems to have a major role in the development of Cheddar and Camembert cheese by synthesizing S-methylthioacetate (most prevalent volatile sulfur compound found in cheese).[9]
Debaryomyces nepalensis
Debaryomyces nepalensis is an osmotolerant yeast isolated from rotten apples that is known to utilize both hexoses and pentoses and produce industrially important metabolites like ethanol, xylitol and arabitol. [10]
Sugar Utilization and Ethanol Creation
| Carbon Source | Carbon Source Consumed (g/L) | Ethanol Created (g/L) |
|---|---|---|
| Sucrose | 82.00 | 9.90 |
| Glucose | 84.75 | 9.05 |
| Arabinose | 86.70 | 2.43 |
| Fructose | 80.40 | 9.84 |
| Glycerol | 50.60 | 0.77 |
General Information
Effect of nitrogen sources
The organism was grown in the presence of different sources of nitrogen like, ammonium sulphate, nitrates and nitrites along with yeast extract and its ability to produce ethanol and arabitol was studied. Among them ammonium sulphate served as the best nitrogen source, whereas, in the presence of nitrites and nitrates, the organism failed to metabolize glucose efficiently. Yeast extract proved to be an integral source of amino acids and other vitamins for growth, without which, the organism had low efficiency for its metabolism. [10]
Bacteria
Leuconostoc
Oenococcus
Oenococcus is a genus of Gram-positive bacteria, placed within the family Leuconostocaceae. The only species in the genus was Oenococcus oeni (which was known as Leuconostoc oeni until 1995). In 2006, the species Oenococcus kitaharae was identified. As its name implies, Oenococcus holds major importance in the field of oenology(the science and study of wine and winemaking), where it is the primary bacterium involved in completing the malolactic fermentation. [11]
Oenococcus kitaharae
O. kitaharae is a lactic acid bacterium (LAB) that was isolated from composting distilled shochu residue produced in Japan. This species represents only the second member of the genus Oenococcus to be identified. O. kitaharae has the ability to ferment maltose, citrate and malate and the ability to synthesize specific amino acids such as L-arginine and L-histidine unlike some O. Oeni. In addition to these metabolic differences, the O. kitaharae genome also encodes many proteins involved in defense against both bacteriophage (restriction-modification and CRISPR) and other microorganisms (bacteriocins), and has had its genome populated by at least two conjugative transposons, which is in contrast to currently available genome sequences of O. oeni which lack the vast majority of these defense proteins. It therefore appears that the genome of O. kitaharae has been shaped by its need to survive in a competitive growth environment that is vastly different from that encountered by O. oeni, where environmental stresses provide the greatest challenge to growth and reproduction. [12]
Sugar Utilization -
One of the defining biochemical differences between O. kitaharae and O. oeni that was noted in its original isolation was the ability of O. kitaharae to produce acid from maltose. This trait is rare in O. oeni, which is formally classified as maltose negative. By comparing available whole-genome annotations for O. oeni with O. kitaharae, it was possible to identify several genes associated with sugar utilization that are deferentially present across the species. Of these, at least four genes which are present in O. kitaharae, but absent in the O. oeni genomes, are predicted to be involved in the utilization of maltose, providing a direct genetic basis for this phenotype. In addition to genes predicted to be involved in the species-specific utilization of maltose, there are several ORFs predicted to be involved in the metabolism of trehalose, D-gluconate, D-ribose and fructose that are specifically present in O. kitaharae. While the assimilation of these sugars is often carried out by specific strains of O. oeni, this genotypic data agrees well with biochemical tests performed previously that indicated that O. kitaharae was able to utilize all of these various carbon sources. [13]
Oenococcus oeni
Oenococcus oeni(also know as Leuconostoc oeni) is a Genus of Gram-positive LAB, ellipsoidal to spherical in shape that is primarily used in Malolactic Fermentation. Oenococcus oeni is a facultative anaerobe. It is able to use oxygen for cellular respiration but can also gain energy through fermentation. It characteristically grows well in the environments of wine, being able to survive in acidic conditions below pH 3.0 and tolerant of ethanol levels above 10%. Optimal growth occurs on sugar and protein rich media. Cells tend to grow in chains or pairs. O. Oeni is heterofermentative and generally produces CO2, Ethanol, Acetate, and Diacetyl. [14]
O. oeni ferments sugars using both the hexose-monophosphate and phosphoketolase pathways using the enzymes Glucose-6-phosphate and xylulose-5-phosphoketolase to from D(-)-lactic acid, CO2 and ethanol in equal amounts when metabolising D-glucose. O.oeni can convert pentose phosphate to acetic acid in an oxygen dependant reaction which requires NADP. It cannot metabolize polysaccharides and alcohols.
O. oeni can decarboxylate L-malate to L(+)-lactate, but cannot use it as a sole source of carbon. It requires the amino acids Glutamic acid, valine, guanine, adenine, xanthine, uracil, riboflavin, folic acid, nicotinic acid, thiamine, biotine and pantothenic acid. There is some variation of amino acid requirement between strains. [15]
Althought O. oeni has primarily been used for Malolactic Fermentation, trials with the White Labs culture(only one reported on so far) has show lactic acid production without the presence of malic acid. James Sites reported souring within a week at 70°F. [16]
| Name | Mfg# |
|---|---|
| White Labs | Malolactic Culture |
| Wyeast | Malolactic Blend |
| CHR Hansen | Viniflora |
Weisella
See also:
Potential references
- Performance of non-conventional yeasts in co-culturewith brewers’ yeast for steering ethanol and aroma production (http://onlinelibrary.wiley.com/doi/10.1111/1751-7915.12717/epdf) - See Fig3B - Fugelsang K, Edwards C. Wine Microbiology. 1997. Available: http://link.springer.com/content/pdf/10.1007/978-0-387-33349-6.pdf - https://www.facebook.com/groups/MilkTheFunk/permalink/1336235339738010/?comment_id=1336277939733750&comment_tracking=%7B%22tn%22%3A%22R%22%7D - https://www.facebook.com/groups/MilkTheFunk/permalink/1337089182985959/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1346900285338182/ - http://www.sciencedirect.com/science/article/pii/S0963996916302332 - https://www.facebook.com/groups/MilkTheFunk/permalink/1366829093345301/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1365795896781954/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1380004022027808/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1284664904895054/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1400174630010747/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1420821137946096/ - http://www.sciencedirect.com/science/article/pii/S074000201630452X - https://www.facebook.com/groups/MilkTheFunk/permalink/1457271340967742/ - Review: Pure non-Saccharomyces starter cultures for beer fermentation with a focus on secondary metabolites and practical applications - https://www.facebook.com/groups/MilkTheFunk/permalink/1485339661494243/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1140282595999953/ - https://www.ncbi.nlm.nih.gov/pubmed/12102552 - https://www.facebook.com/groups/MilkTheFunk/permalink/1546044102090465/ - http://beer.suregork.com/?p=3860 - http://ijs.microbiologyresearch.org/content/journal/ijsem/10.1099/ijsem.0.001607 - https://www.facebook.com/groups/MilkTheFunk/permalink/1582089058485969/ - http://www.mbaa.com/publications/tq/tqPastIssues/2017/Pages/TQ-54-1-0215-01.aspx - http://biorxiv.org/content/early/2017/03/27/121103 - https://www.facebook.com/groups/MilkTheFunk/permalink/1640324282662446/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1659004047461136/ - https://www.facebook.com/groups/MilkTheFunk/permalink/1669790909715783/ - http://onlinelibrary.wiley.com/doi/10.1002/jib.381/full - http://www.asbcnet.org/publications/journal/vol/2017/Pages/ASBCJ-2017-2532-01.aspx - https://www.facebook.com/groups/MilkTheFunk/permalink/1680093658685508/ - http://onlinelibrary.wiley.com/doi/10.1002/yea.3146/abstract - https://mail.google.com/mail/u/1/?ui=2&ik=1b8e47c65b&view=att&th=15c548b66cda8ae6&attid=0.1&disp=safe&zw - https://www.facebook.com/groups/MilkTheFunk/permalink/1649825158379025/ - Pichia - http://www.sciencemag.org/news/2017/07/microbe-new-science-found-self-fermented-beer - Sherry Flor - https://www.facebook.com/groups/MilkTheFunk/permalink/1099692053392341/
See Also
Additional Articles on MTF Wiki
External Resources
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 . Screening for new brewing yeasts in the non-Saccharomyces sector with Torulaspora delbrueckii as model. Maximilian Michel, Jana Kopecká. 2016.
- ↑ Eureka's Blog post about T. Delbruecki, 02/10/2014 .
- ↑ . Wikipedia, Obtained 8/1/17
- ↑ 4.0 4.1 . Pichia k Info. Source: Eureka Brewing Blog.
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3485917/. [Genome Sequence of Pichia kudriavzevii M12, a Potential Producer of Bioethanol and Phytase.]
- ↑ 6.0 6.1 . Identification of Pichia apotheca. Authors: Caiti Smukowski Heil, Joshua N. Burton, Ivan Liachko, Anne Friedrich, Noah A. Hanson, Cody L. Morris, Joseph Schacherer, Jay Shendure, James H. Thomas, Maitreya J. Dunham. 2017.
- ↑ Wikipedia. Debaryomyces. Retrieved 09/03/2015.
- ↑ Wikipedia. Debaryomyces hansenii. Retrieved 09/03/2015.
- ↑ 9.0 9.1 9.2 9.3 . Eureka Blog's Post on D. Hansenii, Retrieved 8/9/2017
- ↑ 10.0 10.1 10.2 . Production of ethanol and arabitol by Debaryomyces nepalensis: influence of process parameters. Himabindu Kumdam, Shweta Narayana Murthy and Sathyanarayana N Gummadi. 2013.
- ↑ .
- ↑ Identifcation of O. Kitaharae, Authors: Akihito Endo1, Sanae Okada1 10/1/2006 .
- ↑ . Functional Divergence in the Genus Oenococcus as Predicted by Genome Sequencing of the Newly-Described Species, Oenococcus kitaharae, Authors Anthony R. Borneman, Jane M. McCarthy, Paul J. Chambers, Eveline J. Bartowsky 01/3/2012 .
- ↑ "Oenococcus oeni". Microbe Wiki. Retrieved 07/20/2017.
- ↑ . UC Davis General Info on O. Oeni (No Date Given) .
- ↑ James Site. Milk The Funk Facebook group. 08/04/2015.